A brain implant leaves the lab for marketability. The global BCI race gets real.
China has approved what the uploaded source describes as the world’s first commercially authorised brain implant designed to help some people with paralysis regain hand movement. The device, from Neuracle Medical Technology, uses a brain-computer interface (BCI) to translate brain-movement intentions into commands for a robotic glove.
That matters because BCIs have spent years living in a strange space between miracle story and moonshot demo. This approval drags the category closer to the real market. It also puts fresh pressure on rivals such as Neuralink and other neurotechnology firms racing to turn brain signals into practical tools for patients, hospitals, and eventually much larger markets.
What’s Happening & Why This Matters
A Commercial BCI for Patients With Paralysis
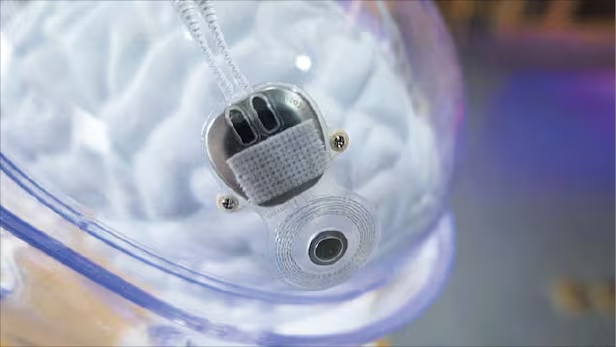
The source says China has approved its first brain implant for commercialisation. It describes the device as the world’s first commercial authorisation for such a product. The implant is designed to help people with paralysis regain some hand movement. The system comes from the Chinese company Neuracle Medical Technology.
That is a notable step because “approved for commercialisation” is not the same as “interesting in a lab.” A commercial approval signals movement from research and pilot use toward real clinical deployment. It suggests regulators believe the system has crossed an important threshold in safety, design, and intended use, even if the eligible patient group is limited.
The target users are also quite specific. According to the file, the system is meant for adults aged 18 to 60 who have had paralysis for more than one year and whose condition remained stable for at least six months. It is intended for people with serious spinal cord injuries in the neck who cannot grip objects with their hands but still retain some movement in their upper arms.
That narrow patient profile matters. It keeps expectations grounded. This is not a universal cure. It is a focused intervention for a defined clinical group.
Using Thoughts to Control a Robotic Glove
The source explains how the device works in plain terms. It detects brain signals tied to the intention to move the hand. Software translates those signals and sends commands to a robotic glove worn by the patient. The glove uses air-powered movement to open and close the hand, allowing the user to grasp objects.

That setup is clever because it does not require the implant to restore the damaged spinal pathway itself. Instead, it creates a bypass. The brain still forms the intention to move. The software reads that intention. The glove then performs the physical motion.
This is one of the most practical versions of BCI logic. It is less about science-fiction mind control and more about functional assistance. Can the user open the hand? Can the user grasp something? Can daily life be a bit less dependent on another person?
Those questions matter more than futuristic hype. A BCI that helps someone perform a useful physical task is much easier to defend clinically than one that makes a good demo clip.
BCI as a Strategic Sector
The file says China has increased investment in BCI technology in recent years. It adds that the country has included BCI as a national strategic priority and plans to feature it in upcoming economic planning as a potential driver of future growth.
The choice shows approval is not an isolated medical decision. It is inside a larger industrial policy initiative. China is not only trying to help patients. It is also trying to build an advanced technology sector that overlaps medical devices, neural interfaces, AI software, and high-value manufacturing.
This is how strategic industries usually form. First comes the research swell. Then a flagship approval is reached. Then the state and private firms start treating the field as a future growth engine.
In that sense, the approval is both a health story and an economic story. It signals that China wants a serious seat at the neurotechnology table, not a spectator pass.
More Entries into the BCI Race
The file places Neuracle’s approval inside a wider international race.
It notes that another Chinese BCI company, Shanghai NeuroXess, drew global attention last year after a 28-year-old man who had been paralysed for eight years after a severe spinal cord injury was able to control digital devices using his thoughts five days after receiving an implant in China.
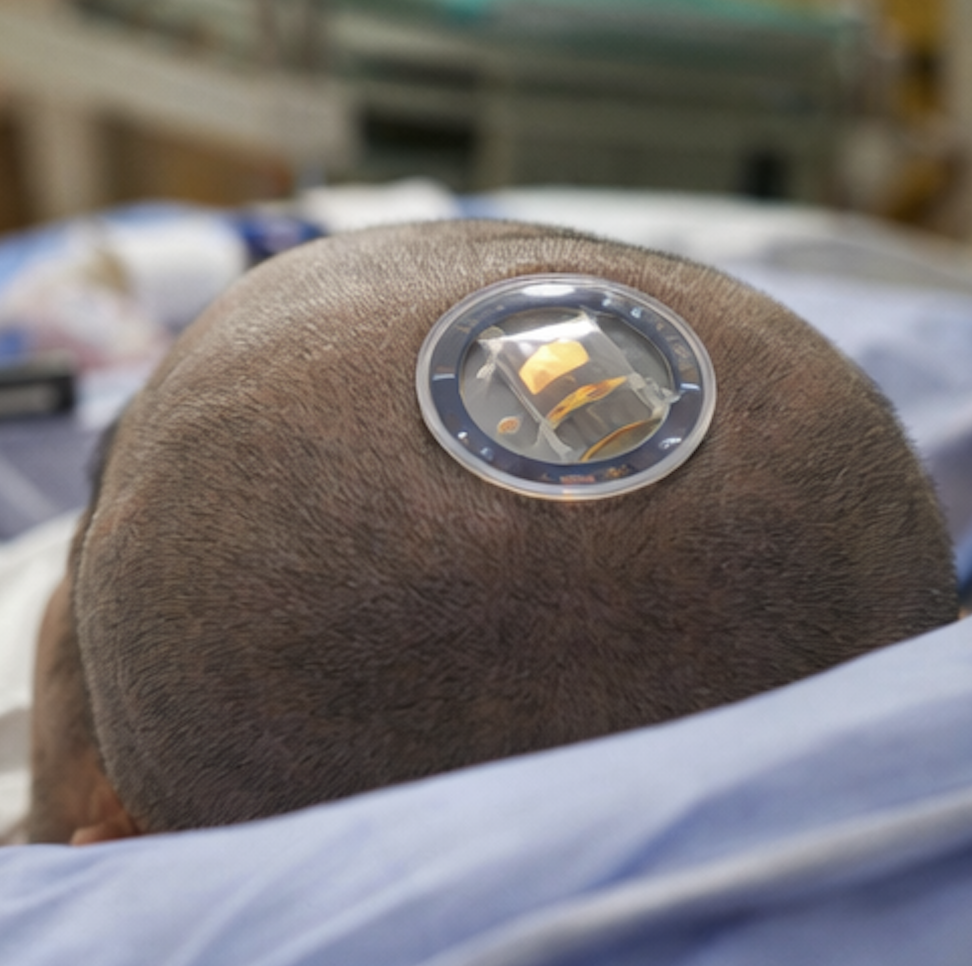
The file also points to Elon Musk’s Neuralink. Musk recently posted on X that Neuralink would begin “high-volume production” of BCI devices in 2026. The source adds that Neuralink said in September last year that 12 people worldwide with severe paralysis had received its implants and were using them to control digital and physical tools through thought.
That comparison is useful because it shows two different races happening at once.
One race is clinical and regulatory. Who gets approved first? Who can show practical value in patients? Who builds trust with regulators and hospitals?
The other race is industrial and symbolic. Who gets to claim leadership in the next big human-machine interface category?
China’s approval gives it a strong talking point in both races. It can say it has moved beyond research attention into commercial authorisation.
The Approval Changes the BCI Market
For years, the BCI field has been heavy on promise and light on routine adoption. Every few months, a company or lab would show a paralysed patient moving a cursor, controlling a digital tool, or manipulating a robotic system with thought. The demonstrations were impressive. They also left the same question hanging in the air: when does any become a product people can actually receive through a structured system?

The approval does not answer every part of that question. It does answer one important piece: at least one BCI system has a commercial path described in the source material.
That changes investor perception. It also changes competitive pressure. Rivals can no longer sell only the romance of the future. They have to respond to a market where regulatory milestones are becoming real and where the first practical systems may start building clinician familiarity before others are ready.
And yes, that is where the strange modern pattern appears again. Once one company or country turns a frontier technology into a product category, everyone else suddenly starts sounding less visionary and more late.
TF Summary: What’s Next
China has approved a brain-computer interface implant from Neuracle Medical Technology for commercialisation, targeting adults aged 18 to 60 with stable paralysis caused by serious neck-level spinal cord injuries. The system reads brain signals associated with hand-movement intention and uses software to drive an air-powered robotic glove that opens and closes the hand. The approval comes as China increases investment in BCI technology and treats the field as a national strategic priority.
MY FORECAST: Neuracle’s approval will send the BCI sector into a more competitive and more practical phase. The next winners will not be decided only by the flashiest demos. They will be judged on regulatory progress, patient usefulness, manufacturability, and clinical trust. China has a strong headline advantage. The bigger battle will be whether that lead turns into real medical adoption before global competitors catch up.
— Text-to-Speech (TTS) provided by gspeech | TechFyle